 We find good agreement with experiment when the mole fraction of water clusters, calculated from a statistical mechanics based theory, is used in the log mixing rule. water, so their greater London forces result in a higher. A possible explanation for failure of the log mixing rule is that the water dissolved in the oil exists not as monomers but as hydrogen-bonded clusters. Although water molecules are much smaller than hexane molecules, water is more viscous than hexane. While a log mixing rule or a one-quarter power mixing rule overestimated viscosity effects, a mole-fraction-weighted average of oil and water viscosities matched the experimental data. The viscosity reduction, although quite significant, was not as pronounced as the drop estimated by viscosity mixing rules used for hydrocarbon systems. At the highest experimental temperature of 286 ☌, viscosities of water-saturated samples were about one-half those of water-free counterparts. That unit does not prove to be convenient for many uses, but note that the. A fluid with low viscosity flows easily because its molecular makeup results in very little friction when it is in motion. A fluidwith large viscosity resists motion because its molecular makeup gives it a lot of internal friction. It describes the internal friction of a moving fluid. Viscosity of liquids is expressed in a variety of units: with the standard SI unit being the Pascal second or Poiseuille. Viscosity is a measure of a fluid's resistance to flow. In this work, the effect of water on viscosity was measured for four crude samples with gravities ranging from 0.97 to 1.03 g/cm 3. The viscosity of water has been studied thoroughly because of the precision with which water flow must be controlled in many industrial applications. The word viscosity comes from the Latin word for mistletoe, viscum. A fluid with a high viscosity, such as honey, flows as a slower rate than a less viscous fluid, such as water. By definition, viscosity is a fluid’s resistance to flow or deformation. At 25 ☌, how high will water rise in a glass capillary tube with an inner diameter of 0.63 mm Refer to Example 10.4 for the required information. Honey, syrup, motor oil, and other liquids that do not flow freely, like those shown in Figure 1, have higher viscosities.  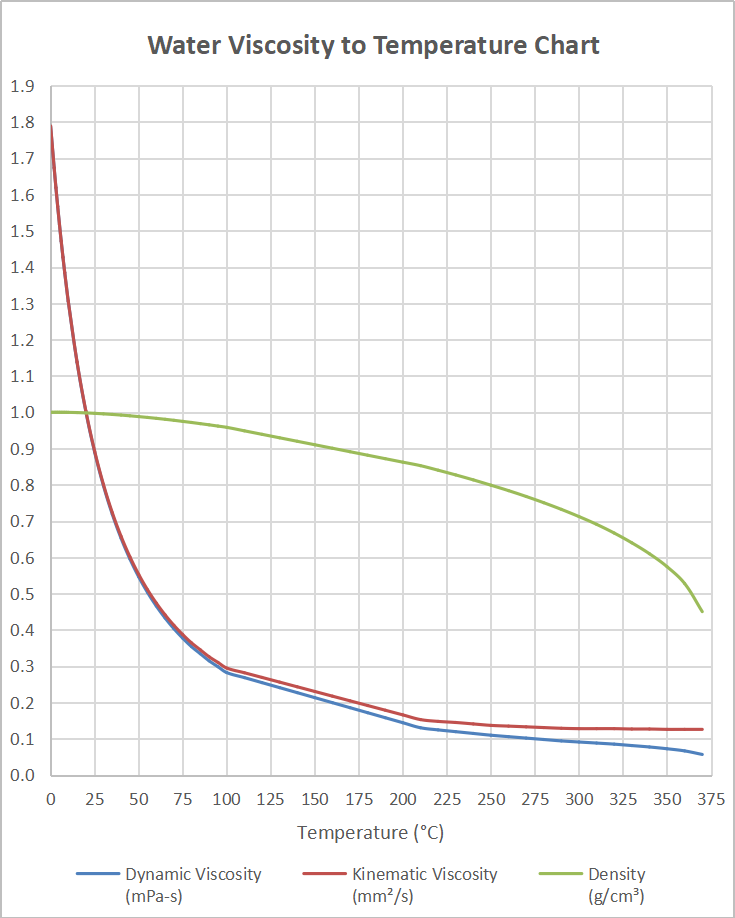
This phenomenon has been considered in thermal recovery simulations but has never been substantiated. Viscosity is a fluid’s resistance to flow. Water, gasoline, and other liquids that flow freely have a low viscosity. 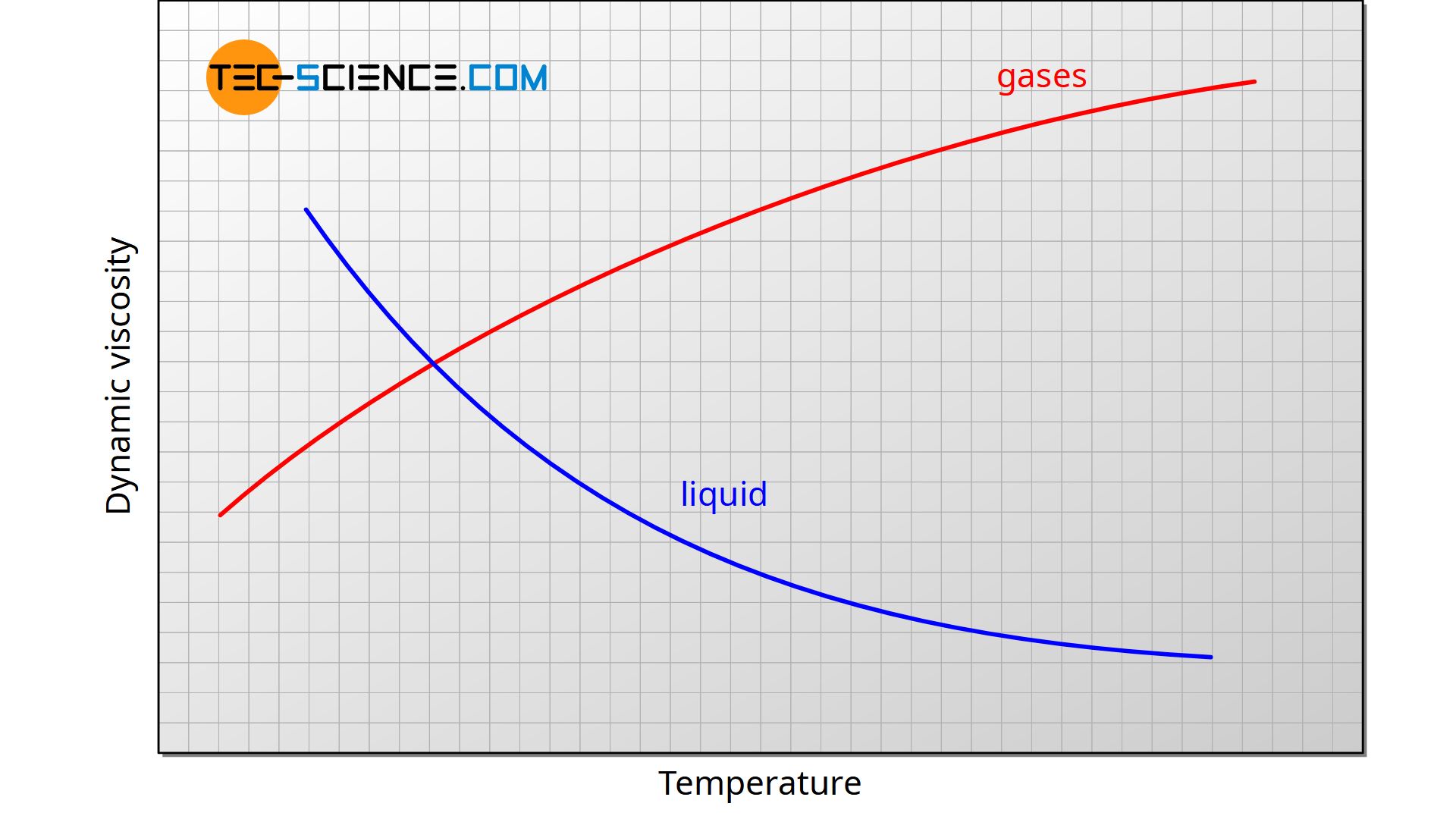
Dissolved water acts as a low-viscosity solvent that reduces oil-phase viscosity. Water dissolution in crude oil becomes significant at temperatures > 150☌, and at 250☌, water solubilities in heavy crudes are ≈ 40 mol%. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
